The development of any new drug or therapy for Rett syndrome or any disorder must follow a scientifically rigorous and well-regulated path. This path is designed to make sure that any new therapy is safe and effective when it is made available to the public. The therapy development path is divided into five stages.
Research Stages
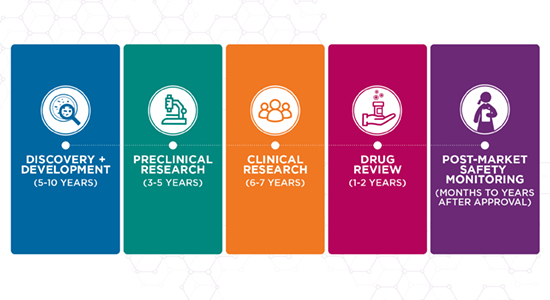
The development of a new therapy for Rett syndrome or any disorder must follow the path that regulatory bodies (such as the U.S. Food and Drug Administration (FDA) or European Medicines Agency) have established. This path helps to assure that any new therapy is safe and effective when it is made available to the public. The therapy development path is divided into five stages. Because of the challenges involved in developing a new therapy, the overall path takes 10-25 years to complete.
Stage 1: Discovery and Development (5-10 years)
The development of a therapy begins in the lab as a basic research discovery. Scientists work for several months to years to generate these discoveries. In this stage, scientists identify new ways to modify a disease process or new disease-fighting technologies. They may identify a new drug, often by testing a library of 100,000+ compounds.
When they find a new treatment approach that looks promising in a first set of experiments, researchers gather more information to develop it. They get this information by performing experiments designed to answer questions about the new therapy, including:
- How does it react with the body?
- Are there any side effects?
- What is the best dose to use?
- Can the new treatment be modified to improve its potential to attain the desired outcomes?
- What is the best way to give the new treatment (such as by mouth or injection)?
- How does the new treatment compare to other treatment approaches to address patients’ health needs?
All these questions must be answered before a new treatment can be developed further. A large number of different experiments are required, which is why this stage of drug development is often one of the longest.
Researchers perform all tests in this stage of drug development in cell culture or animal models. They review all the data they collect. This review gives researchers the information they need to determine whether or not to continue developing the new treatment.
Stage 2: Preclinical Research (3-5 years)
If a new therapy shows promising results in Stage 1, it moves on to the preclinical research stage (Stage 2). Preclinical research carefully tests the new drug or approach for potential to cause severe sickness, injury, or death in cell culture and animal models. This stage follows the regulatory agency mandated requirements. As in Stage 1, many different kinds of experiments are involved. The results of these experiments are carefully reviewed to determine if the new drug or approach is safe enough to test in humans.
Stage 3: Clinical Research (6-7 years)
If the sponsors leading the investigation of a new treatment or approach decide it looks safe and promising in preclinical testing and should be developed further, researchers design experiments to test it in humans. These clinical research designs define:
- Who qualifies as a participant?
- How many people will be involved?
- How long the study will last?
- How bias in the research will be limited?
- What data will be collected?
- How the data will be analyzed?
These trials are necessary to determine if the treatment produces the desired outcome in the human body. To protect human participants as much as possible, the new treatment sponsor must apply to the regulatory authorities for permission to conduct clinical research. In their application, the treatment sponsors must provide basic information about the new treatment (like how it is made), information about study investigators, the data obtained from preclinical testing, and all the details of their clinical research design.
A new treatment can only enter the clinical trial stage if regulatory authorities approve this application. The success of participant recruitment, the length of time participants need to be monitored, and how clear the results are all factors that affect the duration of the clinical research state.
Stage 4: Drug Review (1-2 years)
If the results from pre-clinical and clinical trials indicate that a new treatment approach is safe and effective, the sponsor can apply to the regulatory agency for permission to market it. During this drug review stage, regulatory authorities study all information collected about the new therapy. They then decide whether to approve, reject, or request additional data. If approved, the sponsor develops prescribing information with instructions for using the new treatment, and the treatment enters the market.
Stage 5: Post-Market Safety Monitoring (months to years after approval)
Regulatory agencies continue to monitor the outcomes of patients being treated with a new therapy after its approval. As more people receive the new treatment, more data becomes available. This data may reveal new information about the therapy since it is impossible to find out everything during the therapy development process. Continued safety monitoring enables the prescribing information for a therapy to be changed if needed to protect patients.
Click here to print this information.